Genetics and Evo-Devo of Flower color transitions
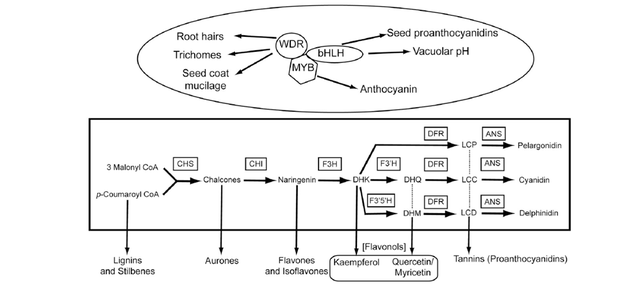
Even though pigmentation traits have had substantial impacts on the field of animal evolutionary developmental biology, they have played only relatively minor roles in plant evo-devo. This is surprising given the often direct connection between flower color and fitness variation mediated through the effects of pollinators. At the same time, ecological and evolutionary genetic studies have utilized the molecular resources available for the anthocyanin pathway to generate several examples of the molecular basis of putatively adaptive transitions in flower color. This area of our research attempts to unite these fields to understand the impact of development on the genetic changes responsible for ecologically important transitions in flower color in nature.
From an ecological genetic perspective, we have worked to characterize the primary genetic changes responsible for the transition from yellow to red flowers between the red and yellow ecotypes of M. aurantiacus in southern California (Streisfeld and Rausher 2009; Streisfeld et al. 2013). In addition, we have identified the causal mutation responsible for a rare, segregating white-flowered polymorphism in the otherwise pink-flowered Mimulus lewisii (Wu et al. 2013). These studies take advantage of classic genetic tools, along with functional and molecular studies of gene expression to test the role of specific candidate genes in flower color transitions.
With these data (and others identified from the literature), we can now begin to test hypotheses about the predictability of the genetic changes responsible for repeated transitions in flower color. Specifically, we have shown that mutations in a single class of transcription factor (the R2R3-Mybs) are over-represented relative to the frequency of observed mutations in other genes that can alter flower color (Streisfeld and Rausher 2011). However, this pattern holds only for mutations that have become fixed between populations or species, but not for those that are segregating within populations (Wu et al. 2013; Sobel and Streisfeld 2013). A leading explanation for this is that deleterious pleiotropic effects of other mutation types are unable to fix in nature, but because of gene duplication and tissue-specificity among R2R3-Myb gene copies, flower-color altering mutations in these genes have fewer negative side effects. We have also explored the consequences of these predictions for evolution based on the phylogenetic history of transitions (Sobel and Streisfeld 2013).
Ongoing studies seek to integrate our knowledge of this pathway and its regulation with the detailed phylogenetic, genetic, genomic, and ecological information available among species in the genus Mimulus (Twyford et al. 2015) to test hypotheses about the molecular evolution and evo-devo of flower color transitions.
From an ecological genetic perspective, we have worked to characterize the primary genetic changes responsible for the transition from yellow to red flowers between the red and yellow ecotypes of M. aurantiacus in southern California (Streisfeld and Rausher 2009; Streisfeld et al. 2013). In addition, we have identified the causal mutation responsible for a rare, segregating white-flowered polymorphism in the otherwise pink-flowered Mimulus lewisii (Wu et al. 2013). These studies take advantage of classic genetic tools, along with functional and molecular studies of gene expression to test the role of specific candidate genes in flower color transitions.
With these data (and others identified from the literature), we can now begin to test hypotheses about the predictability of the genetic changes responsible for repeated transitions in flower color. Specifically, we have shown that mutations in a single class of transcription factor (the R2R3-Mybs) are over-represented relative to the frequency of observed mutations in other genes that can alter flower color (Streisfeld and Rausher 2011). However, this pattern holds only for mutations that have become fixed between populations or species, but not for those that are segregating within populations (Wu et al. 2013; Sobel and Streisfeld 2013). A leading explanation for this is that deleterious pleiotropic effects of other mutation types are unable to fix in nature, but because of gene duplication and tissue-specificity among R2R3-Myb gene copies, flower-color altering mutations in these genes have fewer negative side effects. We have also explored the consequences of these predictions for evolution based on the phylogenetic history of transitions (Sobel and Streisfeld 2013).
Ongoing studies seek to integrate our knowledge of this pathway and its regulation with the detailed phylogenetic, genetic, genomic, and ecological information available among species in the genus Mimulus (Twyford et al. 2015) to test hypotheses about the molecular evolution and evo-devo of flower color transitions.

